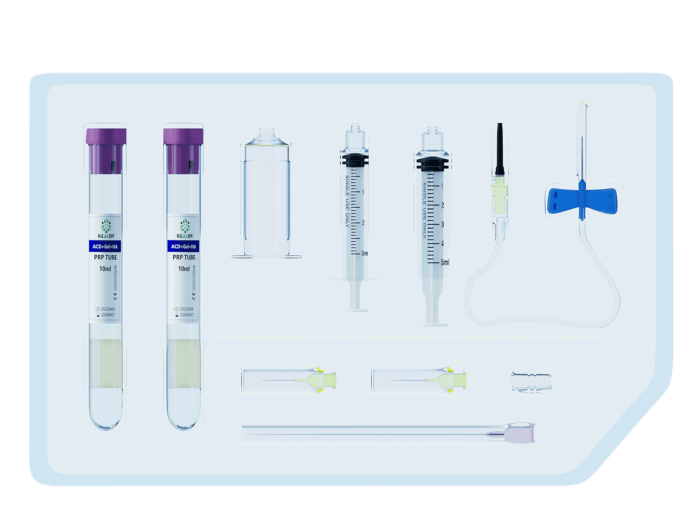
Design points of medical device hard blister box
Packaging of sterile medical device products is a very important and special process that must be strictly controlled in order to ensure the safety and effectiveness of medical device products. Although ISO11607 international standard and EU EN 868-1 standard harmonized to specify the general requirements of packaging materials. In the medical device rigid blister packaging process, the blister packaging design and mold making of the product are particularly important and can directly affect the intended use, failure, transportation and storage of the medical device.
General principles to be considered when designing medical device blister
- Give full play to the characteristics of the plastic itself, such as physical properties, and try to avoid or compensate for its shortcomings. The main considerations are strength, rigidity, toughness, elasticity, water absorption and sensitivity to stress; the molding process of the plastic, such as fluidity; the shrinkage rate after molding and its shrinkage rate of the various anisotropy.
For example, when designing PETG material, the characteristics of the material itself are fully considered. Although PETG has excellent stamping and cutting properties, the stamping force is 60% higher than that of PVC. On the other hand, the cutting time is quite short. The cutting length of a heated die can be increased accordingly, by 11% at 100°C, 25% at 130°C, 40% at 160°C, etc.
- The shape of blister products, under the premise of ensuring the use of the requirements, so that the shape of blister products, simple structure; uniform wall thickness, easy to form and demold; conducive to exhaust, shrinkage and cooling; try to avoid sharp corners (especially the inner corner), to avoid the appearance of various anisotropy of the parts.
- Plastic parts should consider the overall structure of the mold, should fully consider the shape of the subsequent mold and its manufacturing process, have a good economy.
The processability design of medical device blister packaging mainly includes product geometry, dimensional accuracy, stretch ratio, surface roughness, wall thickness, slope, holes, rounded corners, slope of the mold and reinforcement, etc.
- Product geometry and precision. Because thermoforming is a secondary processing, especially differential pressure forming, there is a certain gap and shrinkage deformation between the blister coil and the mold, especially the raised part, the wall becomes thin, so that the strength is affected, so the dimensional accuracy requirements cannot be required too high. (In addition, the product is thin, and the shape and size are very unstable by temperature and environmental changes, so it can’t be treated as precision molding like injection molding).
- Tensile ratio. The ratio of the height (or depth) to the width (or diameter) of the product becomes the stretch ratio. Or called the elongation ratio. Tensile ratio largely reflects the ease of plastic molding, the greater the tensile ratio, the more difficult molding. Generally, the tensile ratio used is 0.5~1.
- Rounded corners. Plastic corner part is not allowed to have sharp corners, corner out of the rounded corners to be as large as possible. The general radius of the corner is not less than the thickness of the processed sheet 4 to 5 times or more, otherwise it is particularly easy to occur in the corner part of the thickness thinning and stress concentration.
- It refers to the slope of the drawing mold, and the slope ranges from 1° to 4°. For negative molding, the lower limit is preferred. This is because the shrinkage of the molded part provides the clearance for attachment.
- Reinforcement bars. Usually in order to ensure the rigidity of the plastic parts, and because the material of the blister product can not be too thick, and also subject to the tensile ratio, the bottom corner part will be thinner, so in the appropriate part of the product design reinforcement, in order to improve the rigidity and strength of the parts.
- Shrinkage of blister products. Blister products have a large post-shrinkage, about 50% of the shrinkage is generated during cooling in the mold. Due to the high mold temperature, the blister products have about 25% shrinkage after cooling to room temperature after demolding, and the remaining 25% is the 24h late cooling. Therefore the design needs to make molds according to different product characteristics.
To sum up, the preliminary design of medical device blister packaging determines the packaging quality and packaging verification of medical devices at the later stage. Although we should consider the packaging requirements of medical device manufacturers, we should pay more attention to the safety and effectiveness of end use.